
The ions align with a polar molecule in such a way that the positive ions are close to the negative part of the dipole and vice versa. Ion-dipole interactions are even stronger than hydrogen bonds. Ion dipole interactions are stronger than the dipole-dipole interactions because an ion has a much stronger charge than a dipole when compared. Ion-dipole interactions are electrostatic forces of attraction between an ion and a polar molecule. Dipole refers to the partial negative and positive charges on a molecule. Ion-dipole interactionsĪn ion is a positively charged (cation) or negatively charged (anion) species. The properties of solids that are determined by intermolecular forces are melting and sublimation. Liquid properties that depend on intermolecular forces include surface tension, viscosity, and diffusion. The gaseous properties that are affected include boiling point, critical point, and vapor pressure. The physical properties of all states of matter are influenced by intermolecular forces. The weakest intermolecular forces are London dispersion forces. These are followed by hydrogen bonds, and then by dipole-dipole interactions. It is because the intramolecular forces hold the atoms in a molecule together whereas intermolecular forces hold different molecules together.Ī comparison of the strength of intermolecular forces against one another would show that ion-dipole interactions are the strongest type of intermolecular force. These electrostatic forces of attraction are present between nuclei and shared electron pairs.Īs a general measure of the strength of intermolecular forces, these are weaker than intramolecular forces (forces within a molecule) such as a metallic bond, ionic bond, or covalent bond. bond length, the intermolecular forces become attractive. In that case, the nuclei of the two atoms, having the same positive charge, repel each other. The forces are repulsive when atoms are very close to each other. These forces can be attractive or repulsive and act between ions or atoms in molecules. As the name suggests, intermolecular forces are the electrostatic forces between molecules. 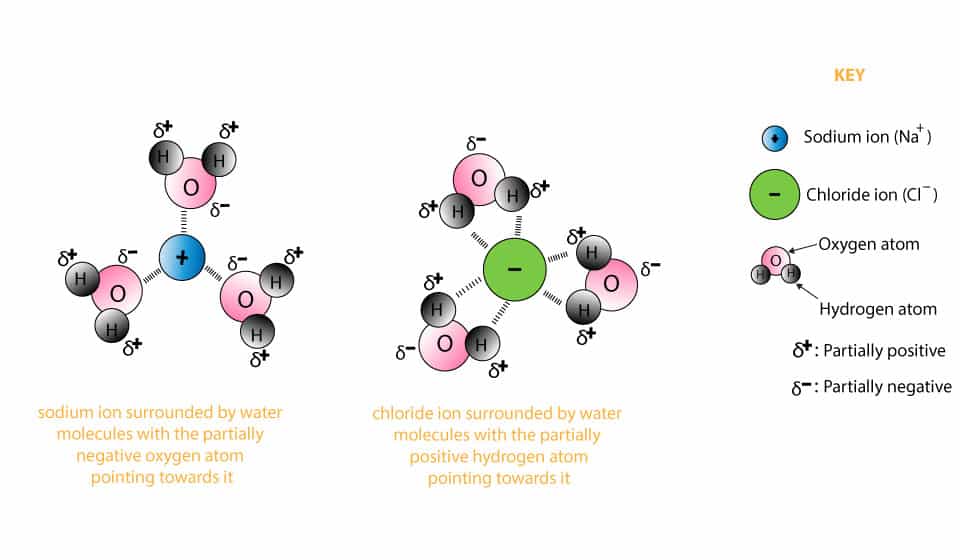
Molecules are formed by the sharing of electrons between atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
